特徴
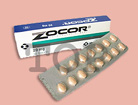
アバンディアは、2型糖尿病の原因の一つであるインスリン抵抗性を直接改善し、内因性のインスリンの作用を高め、高血糖を是正する経口血糖降下剤です。
アバンディアは、1999年5月に米国にて優先審査で承認されて以来、現在までに欧米各国を含む世界101カ国で承認されいます。日本においては、2型糖尿病(単独療法ならびにスルホニル尿素剤との併用)を効能として承認申請中の医薬品です。
Dosage and Administration
The management of antidiabetic therapy should be individualized. Periodic
fasting blood glucose measurements should be performed to monitor
therapeytic response prior to up-titrating dose.
AVANDIA may be taken with or without food.
Monotherapy
The usual starting dose of AVANDIA is 4 mg administered either as
a single dose once daily or in divided doses twice daily. For patiants
who respond inadequately following 12 weeks of treatment as determined
by reduction in FPG, the dose may be increased to 8 mg administered
as a single dose once daily or in divided doses twice daily.
Reductions in glycaemic parameters by dose and regimen are described
under Pharmacodynamics and Clinical Effects. In clinical trials, the
4 mg twice daily regimen resulted in the greatest reduction in FPG
and HbA1c.